endoplasmic reticulum function
Welcome to the solsarin. Let’s read about the post “endoplasmic reticulum function”

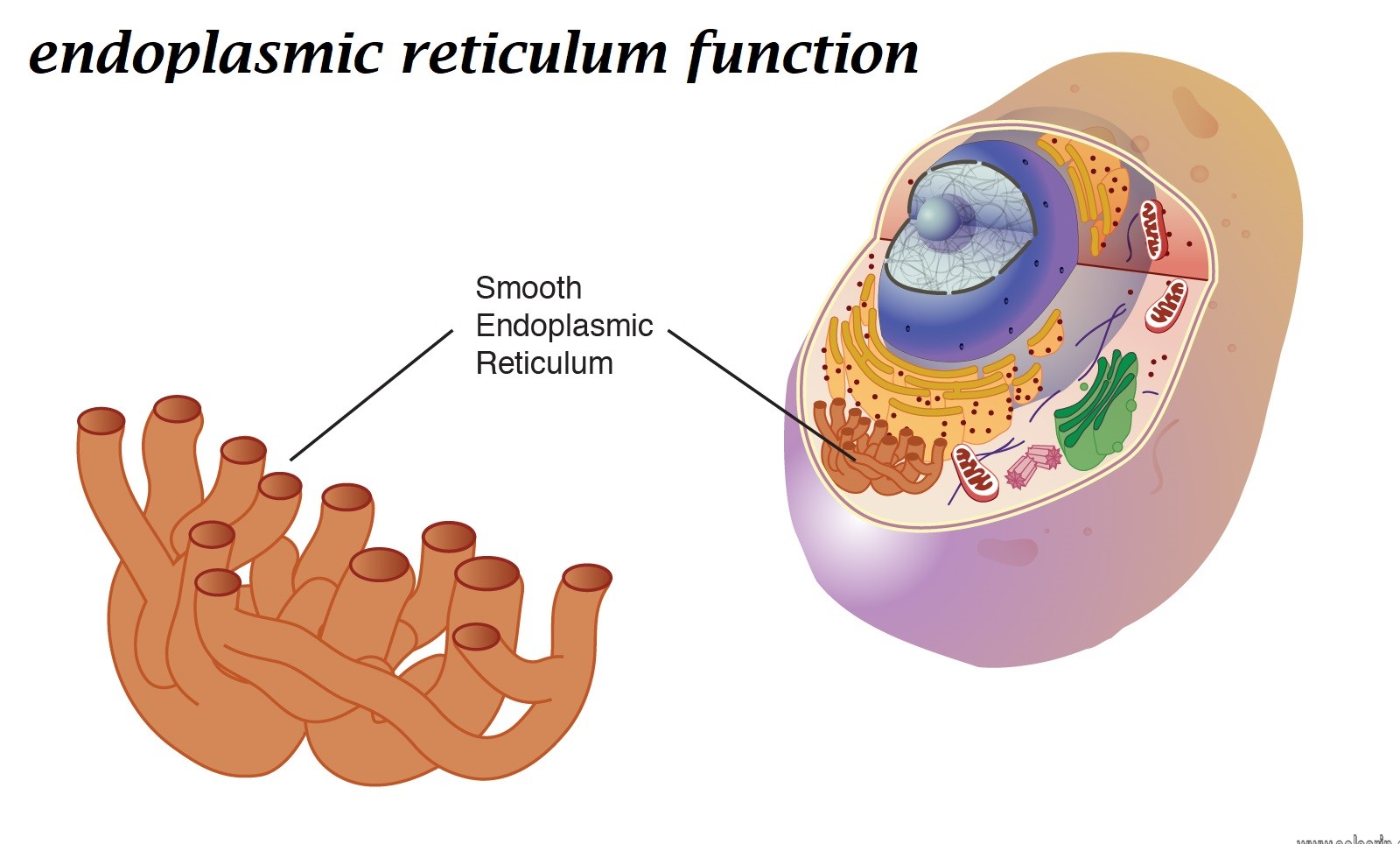
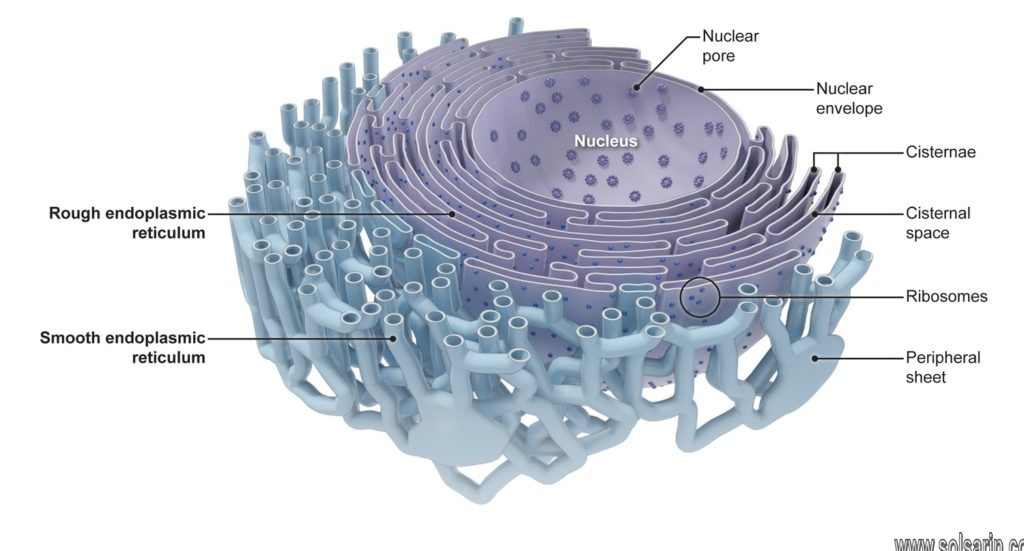
The endoplasmic reticulum (ER) is, in essence, the transportation system of the eukaryotic cell, and has many other important functions such as protein folding. It is a type of organelle made up of two subunits – rough endoplasmic reticulum (RER), and smooth endoplasmic reticulum (SER). The membranes of the ER are continuous with the outer nuclear membrane.
Different types of cells contain different ratios of the two types of ER depending on the activities of the cell.
Rough endoplasmic reticulum(RER) found mainly toward the nucleus of cell and smooth endoplasmic reticulum(SER) found towards the cell membrane or plasma membrane of cell.
The smooth endoplasmic reticulum is especially abundant in mammalian liver and gonad cells.
With electron microscopy, the lacy membranes of the endoplasmic reticulum were first seen in 1945 by Keith R. Porter, Albert Claude, and Ernest F. Fullam. Later, the word reticulum, which means “network”, was applied by Porter in 1953 to describe this fabric of membranes.
Structure
The phospholipid membrane encloses the cisternal space (or lumen), which is continuous with the perinuclear space but separate from the cytosol. The quantity of both rough and smooth endoplasmic reticulum in a cell can slowly interchange from one type to the other, depending on the changing metabolic activities of the cell.
Transformation can include embedding of new proteins in membrane as well as structural changes. Changes in protein content may occur without noticeable structural changes.
Rough endoplasmic reticulum
The binding site of the ribosome on the rough endoplasmic reticulum is the translocon. However, the ribosomes are not a stable part of this organelle’s structure as they are constantly being bound and released from the membrane.
The first 5–30 amino acids polymerized encode a signal peptide, a molecular message that is recognized and bound by a signal recognition particle (SRP).
Translation pauses and the ribosome complex binds to the RER translocon where translation continues with the nascent (new) protein forming into the RER lumen and/or membrane.
The membrane of the rough endoplasmic reticulum forms large double-membrane sheets that are located near, and continuous with, the outer layer of the nuclear envelope.
The double membrane sheets are stacked and connected through several right- or left-handed helical ramps, the “Terasaki ramps”, giving rise to a structure resembling a multi-story car park.
The rough endoplasmic reticulum works in concert with the Golgi complex to target new proteins to their proper destinations.
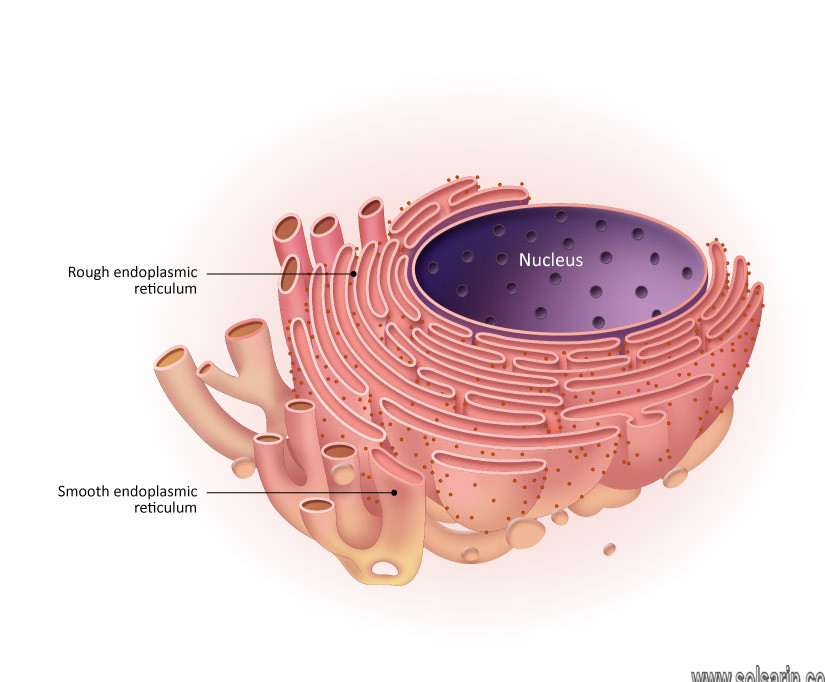

Smooth endoplasmic reticulum
Sarcoplasmic reticulum
The only structural difference between this organelle and the smooth endoplasmic reticulum is the medley of proteins they have. Both bound to their membranes and drifting within the confines of their lumens.
After their release from the sarcoplasmic reticulum, calcium ions interact with contractile proteins. That they utilize ATP to shorten the muscle fiber. The sarcoplasmic reticulum plays a major role in excitation-contraction coupling.
endoplasmic reticulum (ER), in biology, a continuous membrane system that forms a series of flattened sacs within the cytoplasm of eukaryotic cells and serves multiple functions, being important particularly in the synthesis, folding, modification, and transport of proteins .
All eukaryotic cells contain an endoplasmic reticulum (ER). In animal cells, the ER usually constitutes more than half of the membranous content of the cell. Differences in certain physical and functional characteristics distinguish the two types of ER, known as rough ER and smooth ER.
Clinical significance
Increased and supraphysiological ER stress in pancreatic β cells disrupts normal insulin secretion, leading to hyperinsulinemia and consequently peripheral insulin resistance associated with obesity in humans.
Abnormalities in XBP1 lead to a heightened endoplasmic reticulum stress response. And it subsequently causes a higher susceptibility for inflammatory processes that may even contribute to Alzheimer’s disease. In the colon, XBP1 anomalies have been linked to the inflammatory bowel diseases including Crohn’s disease.
The unfolded protein response (UPR) is a cellular stress response related to the endoplasmic reticulum. It is activated in response to an accumulation of unfolded or misfolded proteins in the lumen of the endoplasmic reticulum.
It functions to restore normal function by halting protein translation, degrading misfolded proteins, and activating the signaling pathways. That lead to increasing the production of molecular chaperones involved in protein folding.
Rough ER
It is named for its rough appearance, which is due to the ribosomes attached to its outer (cytoplasmic) surface. It lies immediately adjacent to the cell nucleus.nd its membrane is continuous with the outer membrane of the nuclear envelope.
The proximity of the rough ER to the cell nucleus gives the ER unique control over protein processing. The rough ER can rapidly send signals to the nucleus after that problems in protein synthesis and folding occur. And thereby influences the overall rate of protein translation.
When misfolded or unfolded proteins accumulate in the ER lumen, a signaling mechanism known as the unfolded protein response (UPR) is activated.
The response is adaptive, such that UPR activation triggers reductions in protein synthesis. And enhancements in ER protein-folding capacity and ER-associated protein degradation. If the adaptive response fails, cells are directed to undergo apoptosis (programmed cell death).
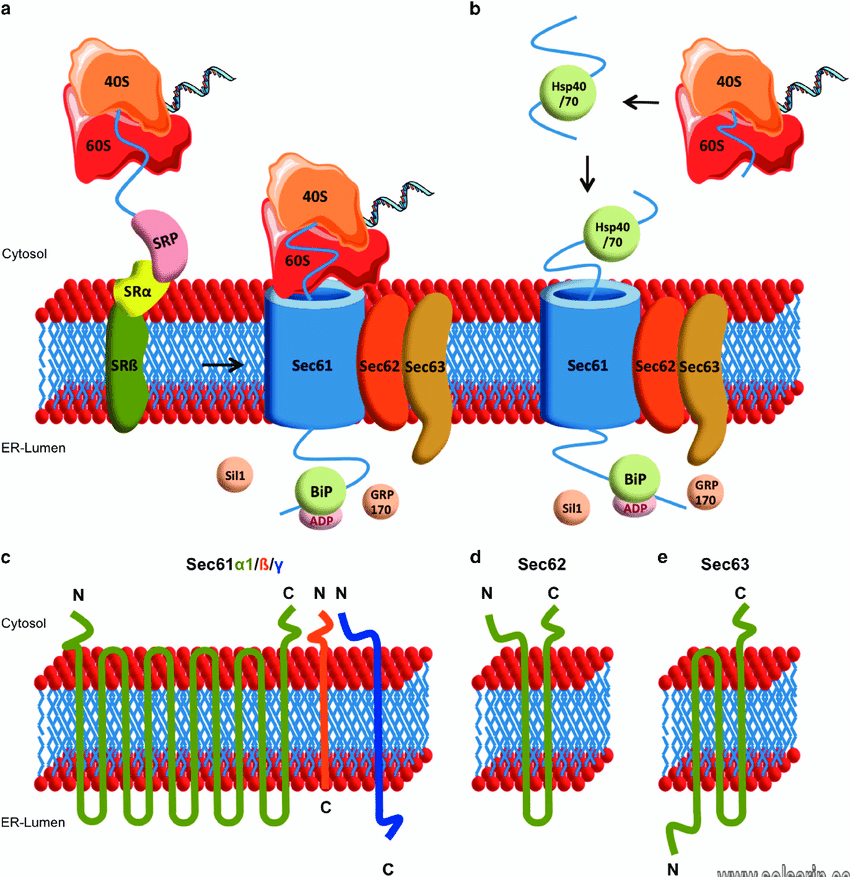

Protein synthesis and folding
One of the major functions of the ER is to serve as a site for protein synthesis for secreted. And integral membrane proteins are, as well as a subpopulation of cytosolic proteins. Protein synthesis requires localization of ribosomes to the cytosolic face of the ER.
And the canonical pathway that regulates protein synthesis involves co-translational docking of the mRNA:ribosome complex on the ER membrane. Translation continues on the ER and the emerging polypeptide can co-translationally enter the ER through the translocon. Which is a channel that contains several Sec proteins and spans the lipid bilayer.
Transmembrane proteins can either contain one hydrophobic stretch of amino acids. And are classified as single pass transmembrane proteins, or contain multiple regions that cross the membrane. And they are classified as multi-pass transmembrane proteins.
Calcium (Ca2+) metabolism
Finally, while the ER is a major site of synthesis and transport of a variety of biomolecules, it is also a major store of intracellular Ca2+. The typical cytosolic concentration of Ca2+ is ~100 nM, while the Ca2+ concentration in the lumen of the ER is 100–800 μM, and the extracellular Ca2+ concentration is ~2 mM.
The ER contains several calcium channels, ryanodine receptors and inositol 1,4,5-trisphosphate (IP3) receptors (IP3R) that are responsible for releasing Ca2+ from the ER into the cytosol when intracellular levels are low.
Ca2+ release occurs when phospholipase C (PLC) is stimulated through G protein-coupled receptor (GPCR) activation and cleaves phosphatidylinositol 4,5 bisphosphate (PIP2) into diacyl-glycerol (DAG) and IP3, which can then bind the IP3R leading to Ca2+ release and transient increase in intracellular Ca2+ levels.
(Ca2+) metabolism
At these regions, clustered STIM1 traps plasma membrane-diffusing Orai1 subunits and assembles them into active Ca2+ release-activated channels (CRAC) allowing for uptake of extracellular Ca2+ into the ER lumen to restore Ca2+ levels.
Release of Ca2+ can result in a wave of Ca2+ that moves through the entire cell, a gradient of Ca2+ from the source of release, or a spatially-restricted wave from clustered channels known as a Ca2+ spark.
Key Takeaways
A cell’s endoplasmic reticulum (ER) contains a network of tubules and flattened sacs. Similarly they performs multiple functions in both plant and animal cells.
Endoplasmic reticulum has two major regions: smooth endoplasmic reticulum and rough endoplasmic reticulum, that contains attached ribosomes.
Via the attached ribosomes, rough endoplasmic reticulum synthesizes proteins via the translation process. It manufactures membranes too.
Smooth endoplasmic reticulum serves as a transitional area for transport vesicles. It functions in carbohydrate and lipid synthesis too. Cholesterol and phospholipids are examples.

Closing remarks
The ER is a complex organelle that plays a pivotal role in protein. And lipid synthesis, calcium storage and stress response. Changes in structure in response to cell cycle or developmental state render this organelle highly dynamic. Several proteins play a role in the proper formation of the different structures including the nuclear envelope, sheets and tubules.
Regulation exists at multiple steps in the formation and maintenance of these structures. And the ratios of these structures are very different in cells of different functions. Or signaling with other organelles would have higher ratios of tubules.
The generation of these structures relies on a myriad of proteins. It involved in either structural aspects of ER morphology by directly affecting the phospholipid bilayer.
And curvature of membranes or mediating interactions with other organelles or the cytoskeleton.
Recent work
Recent work has begun to connect our knowledge of the proteins. That provide the fundamental shape of the ER to signaling pathways.
Recent work on several different human diseases has highlighted a role for several different ER-shaping proteins in diverse diseases.
Thanks for joining this post “endoplasmic reticulum function”.