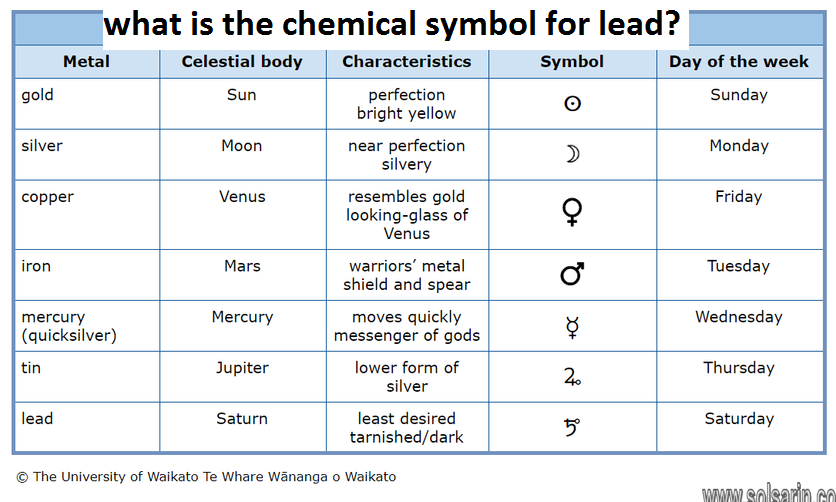
what is the chemical symbol for lead?
Hi everyone, thank you for choosing us. have you ever thought about this question “what is the chemical symbol for lead? ”.
today on the solsarin site we are going to answer this question.
Stay with us.
Thank you for your choice.


lead
- Related Topics:
lead (Pb), a soft, silvery white or grayish metal in Group 14 (IVa) of the periodic table. Lead is very malleable, ductile, and dense and is a poor conductor of electricity. Known in antiquity and believed by the alchemists to be the oldest of metals, lead is highly durable and resistant to corrosion, as is indicated by the continuing use of lead water pipes installed by the ancient Romans. The symbol Pb for lead is an abbreviation of the Latin word for lead, plumbum.
| atomic number | 82 |
|---|---|
| atomic weight | 207.19 |
| melting point | 327.5 °C (621.5 °F) |
| boiling point | 1,744 °C (3,171.2 °F) |
| density | 11.29 gram/cm3 at 20 °C (68 °F) |
| oxidation states | +2, +4 |
| electron configuration | [Xe]4f145d106s26p2 or 1s22s22p63s23p63d104s24p64d104f145s25p65d106s26p2 |
Occurrence and distribution
Lead is mentioned often in early biblical accounts. The Babylonians used the metal as plates on which to record inscriptions. The Romans used it for tablets, water pipes, coins, and even cooking utensils; indeed, as a result of the last use, lead poisoning was recognized in the time of Augustus Caesar. The compound known as white lead was apparently prepared as a decorative pigment at least as early as 200 BCE. Modern developments date to the exploitation in the late 1700s of deposits in the Missouri-Kansas-Oklahoma area in the United States.


On a weight basis, lead has nearly the same abundance in Earth’s crust as tin. Cosmically, there is 0.47 lead atom per 106 silicon atoms. The cosmic abundance is comparable to those of cesium, praseodymium, hafnium, and tungsten, each of which is regarded as a reasonably scarce element.
Although lead is not abundant, natural concentration processes have resulted in substantial deposits of commercial significance, particularly in the United States but also in Canada, Australia, Spain, Germany, Africa, and South America. Significant deposits are found in the United States in the western states and the Mississippi valley. Rarely found free in nature, lead is present in several minerals, but all are of minor significance except the sulfide, PbS (galena, or lead glance), which is the major source of lead production throughout the world. Lead is also found in anglesite (PbSO4) and cerussite (PbCO3). By the early 21st century, China, Australia, the United States, Peru, Mexico, and India were the world’s top producers of lead in concentrate.
Additional refining removes impurities present in the lead bullion produced by either process.
Uses of the metal
Properties that are responsible for the many uses of elemental lead include its ductility, ease of welding, low melting point, high density, and ability to absorb gamma radiation and X-radiation. Molten lead is an excellent solvent and collector for elemental silver and gold. The structural applications of lead are limited by its low tensile and fatigue strengths and its tendency to flow even when only lightly loaded.
When freshly cut, lead oxidizes quickly, forming a dull gray coating, formerly thought to be lead suboxide, Pb2O, but now recognized as a mixture of lead and lead monoxide, PbO, which protects the metal from further corrosion. Similarly, although lead is soluble in dilute nitric acid, it is only superficially attacked by hydrochloric or sulfuric acids because the insoluble chloride (PbCl2) or sulfate (PbSO4) coatings that are formed prevent continued reaction.
Elemental lead can also be oxidized to the Pb2+ ion by hydrogen ions, but the insolubility of most salts of Pb2+ makes lead resistant to attack by many acids. Oxidation under alkaline conditions is easier to effect and is favoured by the formation of the soluble species of lead in the +2 oxidation state. Lead oxide (PbO2, with lead as the Pb4+ ion) is among the stronger oxidizing agents in acidic solution, but it is comparatively weak in alkaline solution.
Lead has many other applications, the largest of which is in the manufacture of storage batteries.
Properties of the element
Lead and its compounds are toxic and are retained by the body, accumulating over a long period of time—a phenomenon known as cumulative poisoning—until a lethal quantity is reached. The toxicity of lead compounds increases as their solubility increases. In children the accumulation of lead may result in cognitive deficits; in adults it may produce progressive renal disease. Symptoms of lead poisoning include abdominal pain and diarrhea followed by constipation, nausea, vomiting, dizziness, headache, and general weakness. Elimination of contact with a lead source is normally sufficient to effect a cure.
The elimination of lead from insecticides and paint pigments and the use of respirators and other protective devices in areas of exposure have reduced lead poisoning materially. The recognition that the use of tetraethyl lead, Pb(C2H5)4, as an antiknock additive in gasoline was polluting the air and water led to the compound’s elimination as a gasoline constituent in the 1980s. (For full treatment of lead and lead mining and refining, see also lead poisoning.)
Nuclear properties
Lead is formed both by neutron-absorption processes and the decay of radionuclides of heavier elements. Lead has four stable isotopes; their relative abundances are lead-204, 1.48 percent; lead-206, 23.6 percent; lead-207, 22.6 percent; and lead-208, 52.3 percent. Three stable lead nuclides are the end products of radioactive decay in the three natural decay series:
uranium (decays to lead-206), thorium (decays to lead-208), and actinium (decays to lead-207). More than 30 radioactive isotopes have been reported. Of the radioactive isotopes of lead, the following appear as members of the three natural decay series: (1) thorium series: lead-212; (2) uranium series: lead-214 and lead-210; (3) actinium series: lead-211. The atomic weight of natural lead varies from source to source, depending on its origin by heavier element decay.
Compounds


Lead shows oxidation states of +2 and +4 in its compounds. Among the many important lead compounds are the oxides: lead monoxide, PbO, in which lead is in the +2 state; lead dioxide, PbO2, in which lead is in the +4 state; and trilead tetroxide, Pb3O4. Lead monoxide exists in two modifications, litharge and massicot.
Litharge, or alpha lead monoxide, is a red or reddish yellow solid, has a tetragonal crystal structure, and is the stable form at temperatures below 488 °C (910 °F). Massicot, or beta lead monoxide, is a yellow solid and has an orthorhombic crystal structure; it is the stable form above 488 °C. Both forms are insoluble in water but dissolve in acids to form salts containing the Pb2+ ion or in alkalies to form plumbites, which have the PbO22− ion.High-quality glassware (lead crystal) contains as much as 30 percent litharge, which increases the refractive index of the glass and makes it brilliant, strong, and resonant.
It decomposes upon heating and yields oxygen and lower oxides of lead. PbO2 is used as an oxidizing agent in the production of dyestuffs, chemicals, pyrotechnics, and matches and as a curing agent for polysulfide rubbers. Trilead tetroxide (known as red lead, or minium) is produced by further oxidation of PbO. It is the orange-red to brick-red pigment commonly used in corrosion-resistant paints for exposed iron and steel. It also reacts with ferric oxide to form a ferrite used in making permanent magnets.
Another economically significant compound of lead in the +2 oxidation state is lead acetate, Pb(C2H3O2)2, a water-soluble salt made by dissolving litharge in concentrated acetic acid. The common form, the trihydrate, Pb(C2H3O2)2·3H2O, called sugar of lead, is used as a mordant in dyeing and as a drier in certain paints. In addition, it is utilized in the production of other lead compounds and in gold cyanidation plants, where it primarily serves to precipitate soluble sulfides from solution as PbS.
Various other salts, most notably basic lead carbonate, basic lead sulfate, and basic lead silicate, were once widely employed as pigments for white exterior paints. Since the mid-20th century, however, the use of such so-called white lead pigments has decreased substantially because of a concern over their toxicity and attendant hazard to human health.
what is the chemical symbol for lead chromate
Chrome yellow is lead chromate (PbCrO4). Lead oxide chromate (Pb2O·CrO4) is orange-red. Actually, lead chromate may exist as a lemon yellow rhombic form, a reddish yellow monoclinic form, and a scarlet tetragonal form, but only the monoclinic form is stable at room temperature. Chrome orange and chrome red (PbCrO4·PbO) are produced by heating lead chromate with alkali. The difference in the two colors is related to particle size. Chrome green is a mixture of chrome yellow and Prussian blue. Zinc yellow is zinc chromate loosely combined with zinc hydroxide.
Random Posts
what is the chemical formula for lead(iv) oxide
Lead IV Oxide is a hexagonal dark brown crystalline powder that is insoluble in water. This chemical is widely used in the manufacturing of matches, dyes, electrodes, pyrotechnics, positive plates of lead-acid batteries and explosives. It is an extremely strong oxidant and is harmful and poisonous in nature.
Properties Of Lead IV Oxide
| Chemical formula | PbO2 |
| Chemical names | Plumbic oxide
Plattnerite |
| Molecular weight | 239.198 g/mol |
| Density | 9.38 g/cm3 |
| Melting point | 290 °C |
To form each Pb4+ it requires two negative oxide ions O2− to balance out total positive and total negative charge on it.