is iodine soluble in water or oil
Hello. Welcome to solsarin. This post is about “is iodine soluble in water or oil“.
Iodine
Iodine is a chemical element with the symbol I and atomic number 53. The heaviest of the stable halogens, it exists as a semi-lustrous, non-metallic solid at standard conditions that melts to form a deep violet liquid at 114 degrees Celsius, and boils to a violet gas at 184 degrees Celsius. The element was discovered by the French chemist Bernard Courtois in 1811, and was named two years later by Joseph Louis Gay-Lussac, after the Ancient Greek Ιώδης ‘violet-coloured’.
Iodine occurs in many oxidation states, including iodide (I−), iodate (IO−
3), and the various periodate anions. It is the least abundant of the stable halogens, being the sixty-first most abundant element. It is the heaviest essential mineral nutrient. Iodine is essential in the synthesis of thyroid hormones.[4] Iodine deficiency affects about two billion people and is the leading preventable cause of intellectual disabilities.[5]
The dominant producers of iodine today are Chile and Japan. Iodine and its compounds are primarily used in nutrition. Due to its high atomic number and ease of attachment to organic compounds, it has also found favour as a non-toxic radiocontrast material. Because of the specificity of its uptake by the human body, radioactive isotopes of iodine can also be used to treat thyroid cancer. Iodine is also used as a catalyst in the industrial production of acetic acid and some polymers.
It is on the World Health Organization’s List of Essential Medicines.[6]
DEEP PURPLE MAGIC
In this activity, students see colours change in a solution, and learn how chemistry can explain this magical result.
This trick works because iodine is brown when dissolved in water and purple when dissolved in oil.


Pure iodine is violet, but when it’s dissolved in water, it accepts an electron from the oxygen atom, affecting how it absorbs light. When you shake the fluids, the iodine leaves the water and dissolves in the oil, and returns to its purple colour!
The reason the iodine molecules leave water to dissolve in oil is due to how polarity affects solubility. Water is polar; it has an uneven distribution of electrons. Oil is non-polar; electrons in molecules of this substance are distributed evenly. Iodine is also a non-polar molecule therefore it is more soluble in oil; “like dissolves like”.
What To Do
Hint: perform the “magic trick” before you offer an explanation.
- Fill the jar about ½ full of water and add iodine until it is about the colour of tea.
- Hold it up and slowly add an equal amount of oil so that the students can see the clear oil on top of the amber-coloured water.
- Tighten the cap and shake for about 30 seconds. The two layers will need a few moments to form. The bottom layer will be much lighter and the top will be a beautiful purple colour.
Vocabulary:
- Solution: A mixture of two or more substances, usually in liquid form.
- Polar molecule: a molecule that has an uneven distribution of molecules from one end to the other.
Extensions
- Can you predict what would happen if you didn’t have equal amounts of oil and water in the bottle? Test it and see if your results match your predictions.
- Take a slice of potato and add a drop or two of iodine onto it. What colour change do you observe?
- Investigate what is in the potato that causes the colour change when iodine is applied.
Is iodine soluble in water?
Iodine dissolves easily in chloroform and hexane but does not dissolve in water. … Although non-polar molecular iodine cannot dissolve in water, it reacts with iodide ion to form something that can: the triiodide ion.
Does iodine dissolve in water?
Explanation: Iodine is soluble in both ethanol and hexane. Iodine does not dissolve in water because water is an extremely polar molecule, while iodine exists in the diatomic form of I2 , and is therefore non-polar, and will not dissolve in water.
What happens when you mix iodine and water?
Pure iodine is violet, but when it’s dissolved in water, it accepts an electron from the oxygen atom, affecting how it absorbs light. When you shake the fluids, the iodine leaves the water and dissolves in the oil, and returns to its purple colour!
Why is iodine soluble in CCl4?
Iodine is more soluble in CCl4 but less in H2O because Iodine is nonpolar and CCl4 is also non-polar, so like dissolve in like whereas water is polar so iodine is less soluble in H2O.
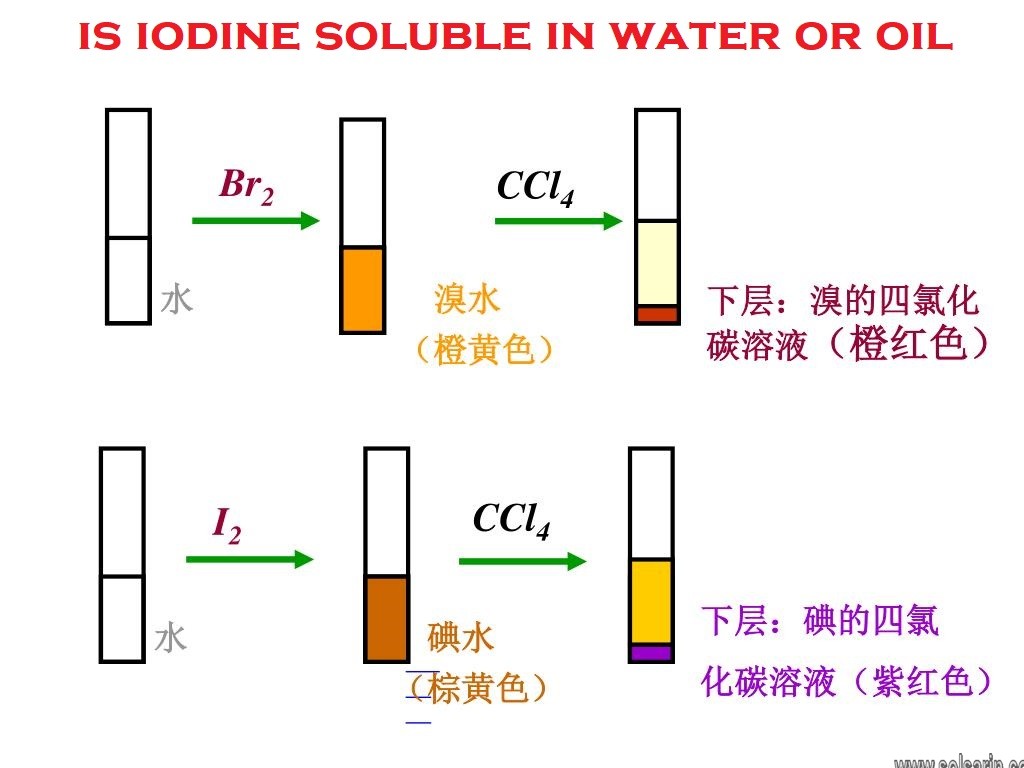
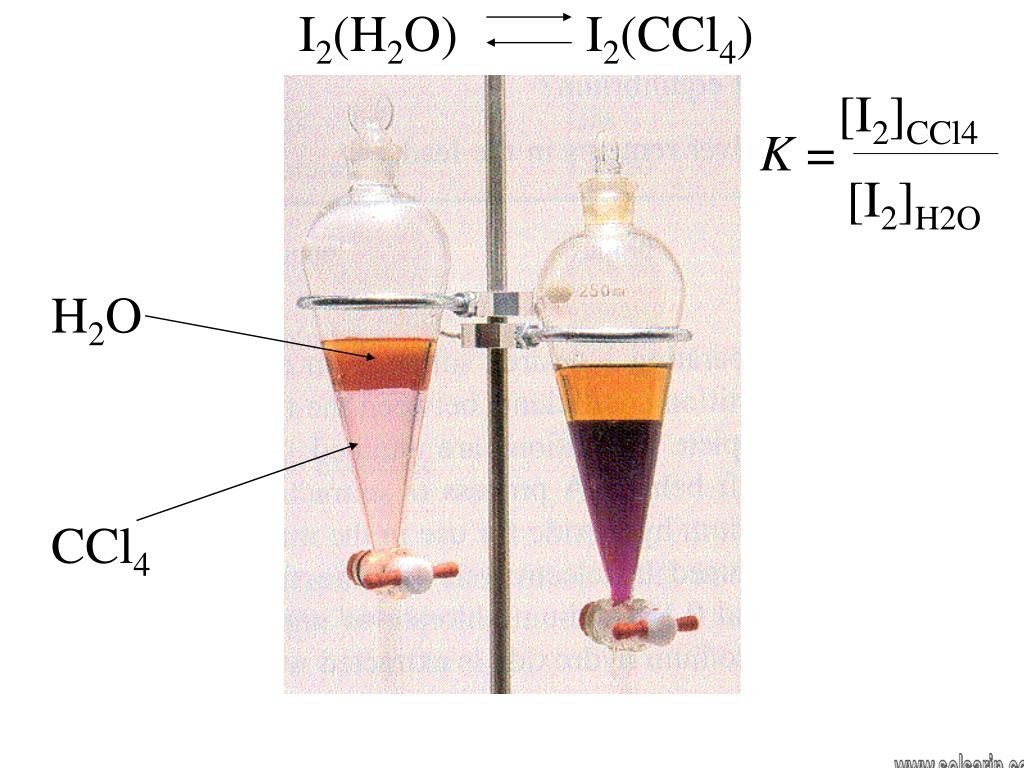
Does iodine dissolve in CCl4?
The I2 molecules in iodine and the CCl4 molecules in carbon tetrachloride are both held together by weak intermolecular bonds. Similar intermolecular bonds can form between I2 and CCl4 molecules in a solution of I2 in CCL4. I2 therefore readily dissolves in CCl4.
Does iodine dissolve better in oil or water?
The reason the iodine molecules leave water to dissolve in oil is due to how polarity affects solubility. Oil is non-polar; electrons in molecules of this substance are distributed evenly. Iodine is also a non-polar molecule therefore it is more soluble in oil; “like dissolves like”.
Why is iodine in water brown?
Why is iodine Brown in water? Iodine is non-polar and thus dissolves in methylene chloride by dispersion forces (induced dipole/induced dipole). A small amount of iodine may dissolve in the water initially due to dipole/induced-dipole interactions producing a slight brown color.

Why is iodine not very soluble in water?
Non-polar Iodine is not very soluble in water. An intermolecular bond between an induced dipole (I2) and a polar bond in water is not very strong compared to the hydrogen bonds in water. The water molecules would rather remain hydrogen bonded to each other, then to allow an iodine molecule come between them.
What Colour is iodine solution?
Iodine Test
A solution of iodine (I2) and potassium iodide (KI) in water has a light orange-brown color.
What is the color when iodine is dissolved in CCl4?
The colour that results is violet.
Why is iodine soluble in water?
Likewise, people ask, why does iodine dissolve in water?
Iodine has a very low solubility in water because it is non-polar. As a result, an I2 molecule cannot form strong interactions with water molecules (no hydrogen bonds or permanent dipole-dipole interactions), whereas water molecules do form strong interactions with each other.
One may also ask, does iodine crystals dissolve in water? Iodine does not dissolve in water, but it does dissolve in a solution of an iodide in water. It dissolves easily in organic solvents.
is iodine soluble or insoluble?
Elemental iodine is slightly soluble in water, with one gram dissolving in 3450 ml at 20 °C and 1280 ml at 50 °C; potassium iodide may be added to increase solubility via formation of triiodide ions, among other polyiodides. Nonpolar solvents such as hexane and carbon tetrachloride provide a higher solubility.
How does iodine react with water?
Iodine reacts with water to some extent to give a mixture of hydroiodic acid and iodic(I) acid – also known as hypoiodous acid. The reaction is reversible, and at any one time only about 0.05% of the iodine molecules have actually reacted and others remain as molecules.
Is Iodine Solution And Water Soluble Or Insoluble
Elemental iodine is slightly soluble in water, with one gram dissolving in 3450 ml at 20 °C and 1280 ml at 50 °C; potassium iodide may be added to increase solubility via formation of triiodide ions, among other polyiodides. Nonpolar solvents such as hexane and carbon tetrachloride provide a higher solubility.
Is iodine solution soluble?
It dissolves readily in many organic solvents, producing violet solutions in most cases and yellow-brown solutions in alcohols. Iodine is not very soluble in water, but produces a slightly yellow solution. No ions are produced when iodine dissolves in any of these solvents.
Why is iodine soluble in water?
So, it is soluble in water. Iodine can be solubilized in water by dissolving iodine in KI and then into the water. As two iodine atoms share two electrons to form a bond, so iodine is a covalent molecule. Iodine accepts the electrons from another molecule in its antibonding orbital.


Does iodine solution mixed with water?
Pure iodine is violet, but when it’s dissolved in water, it accepts an electron from the oxygen atom, affecting how it absorbs light. When you shake the fluids, the iodine leaves the water and dissolves in the oil, and returns to its purple colour!.
What can dissolve iodine?
Iodine is only slightly soluble in water but dissolves readily in a solution of sodium or potassium iodide. Tincture of iodine is a solution of iodine and potassium iodide in alcohol. Iodine also dissolves in carbon disulfide, carbon tetrachloride, and chloroform, giving a deep violet solution.
Thank you for staying with this post “is iodine soluble in water or oil” until the end.